Certified translations that protect your reputation and operations
The first Central American company certified under the ISO 9001 and ISO 17100 standards, specializing in regulated sectors such as medical devices, legal, financial, and sanitary registration.


The reasons why leading companies trust in Tilde Traducciones
Compliance with international standards
Certified translations that avoid regulatory sanctions and critical audit errors.
Reliable and prioritized deliveries
Processes that adapt to your deadlines and phased releases without compromising quality.
Single point of contact
Centralized project management for peace of mind and full traceability.
Beyond translations, we forge trust
Leading companies rely on Tilde as a strategic partner in Free Trade Zones to meet their regulatory and operational goals.
“Thanks to Tilde, our translations were ready for the audit without a hitch. Their project management was impeccable.”
Quality Engineer, Pharmaceutical Company
“We found in Tilde a partner that understands our regulatory processes and knows how to respond at the speed we need.”
Quality Manager, Medical device company
“Their ISO 17100 certification gave us confidence to delegate critical documentation. They met deadlines and ensured terminology consistency.”
Regulatory Affairs Manager, Medical Technology Company
How we do it
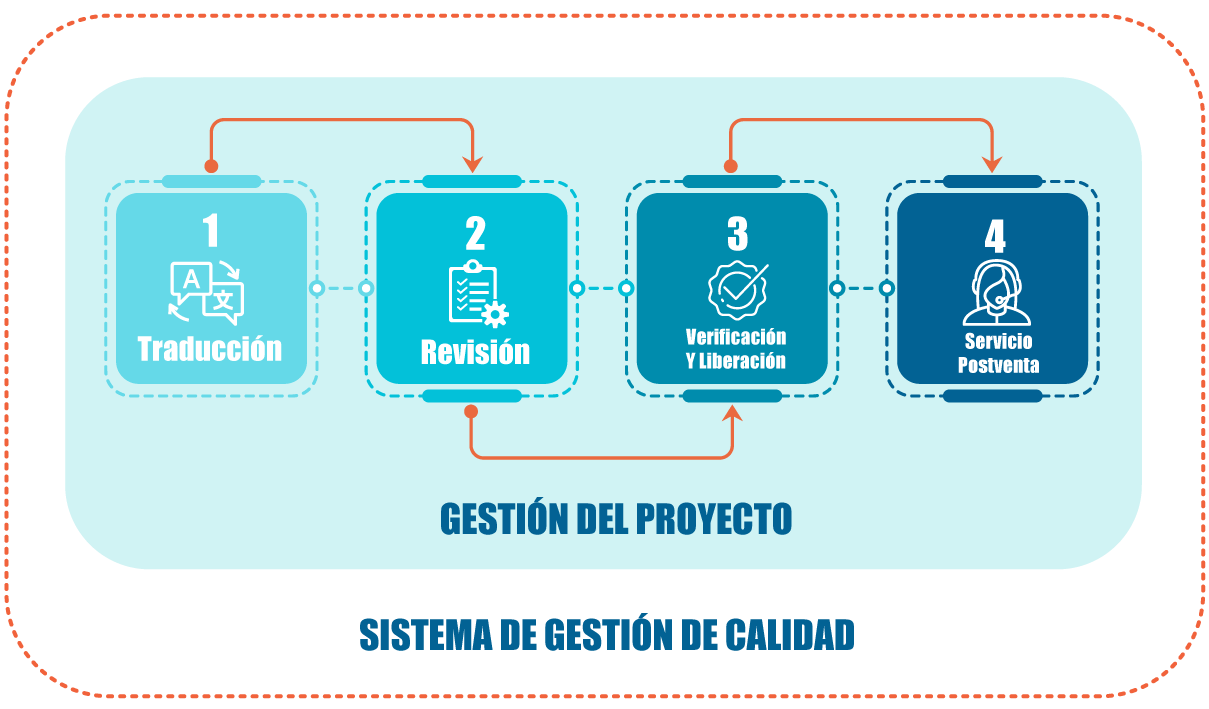
Each project is managed under ISO 9001 and ISO 17100 standards, with professional review, traceability, and quality controls at every stage.
Looking for reliable translations for your company?
Fill in the form and our team will send you a customized quote in less than 24 hours.

